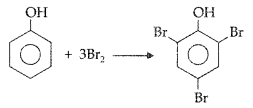
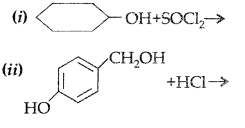
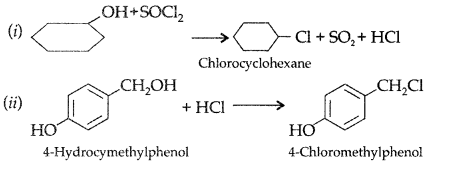
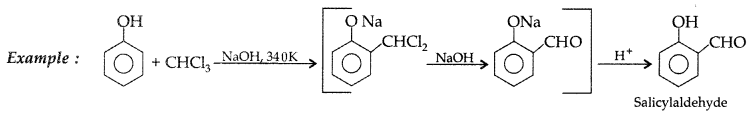
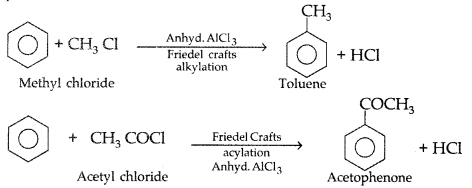
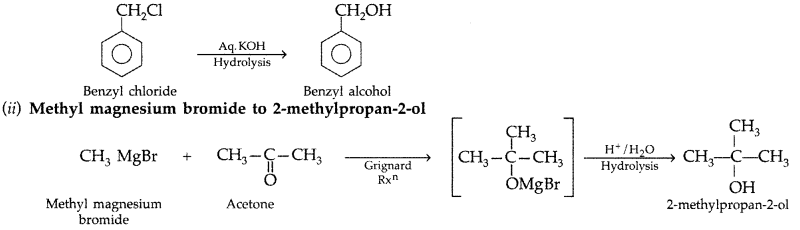
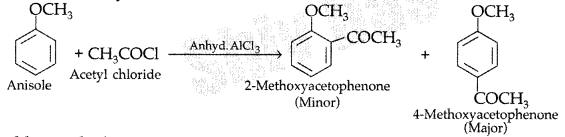
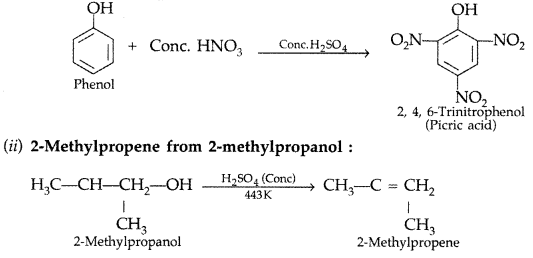
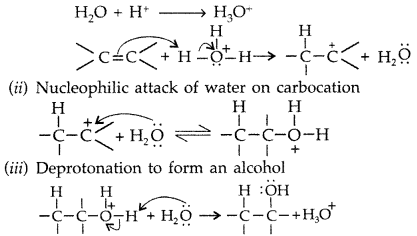
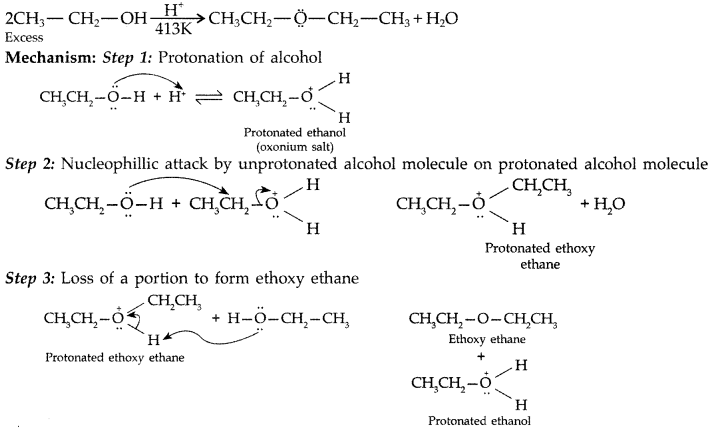
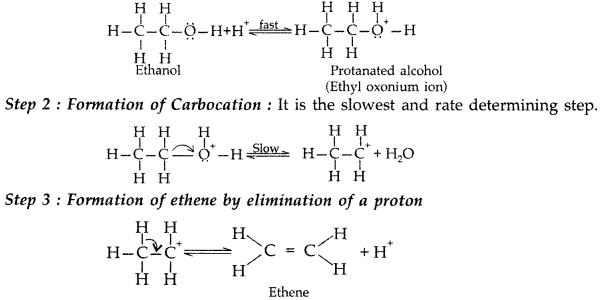
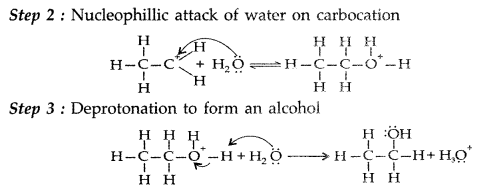
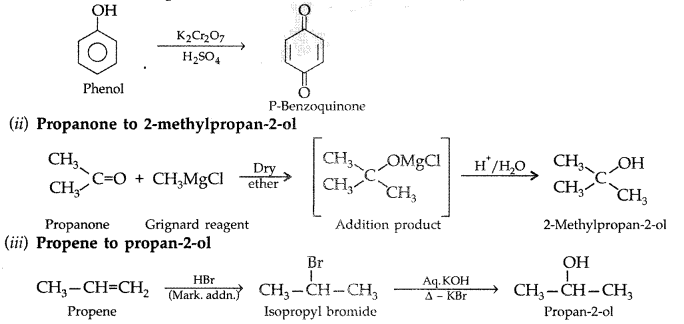
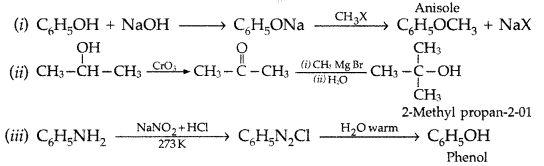
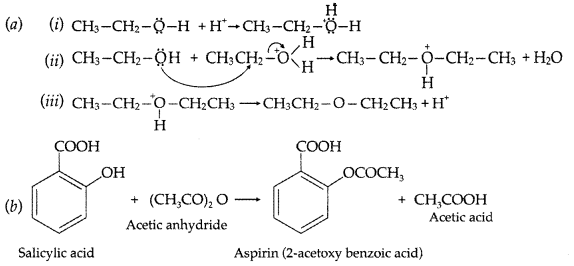
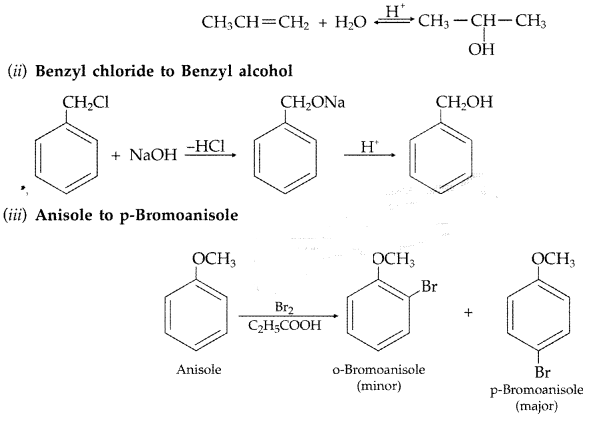
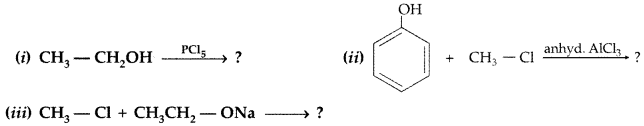
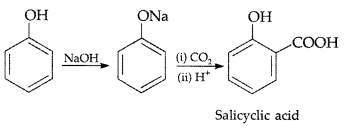
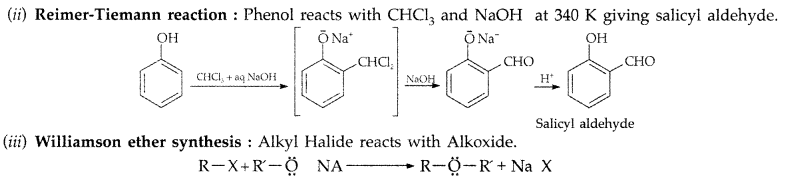
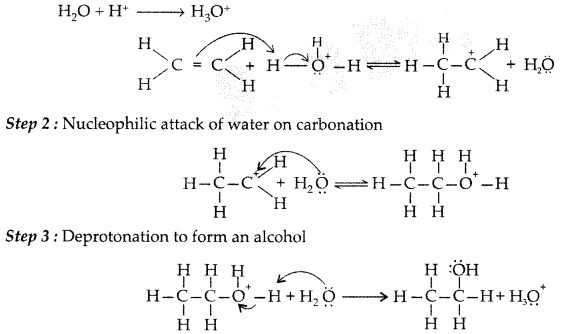
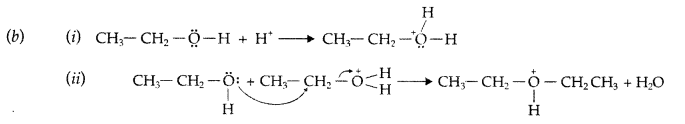
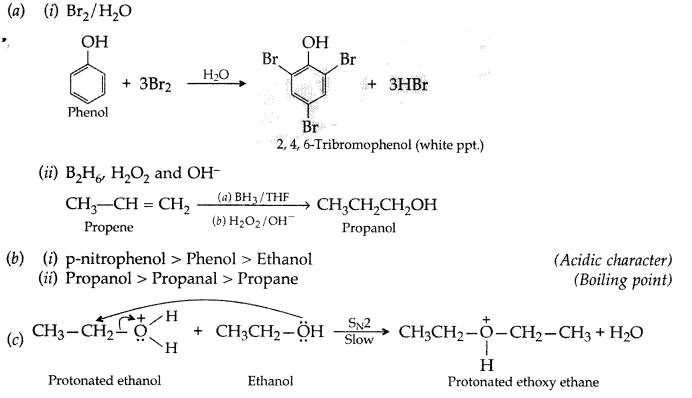
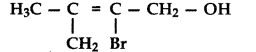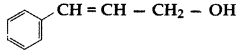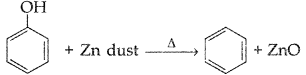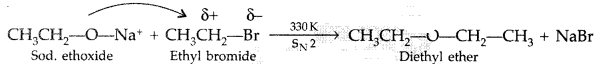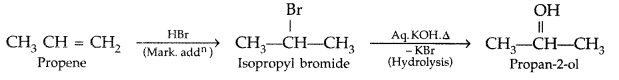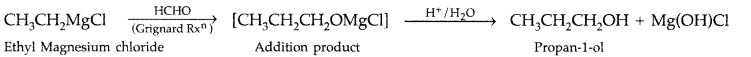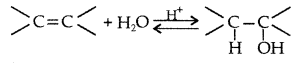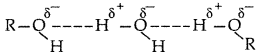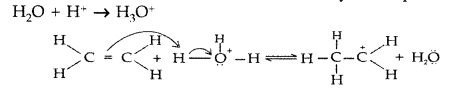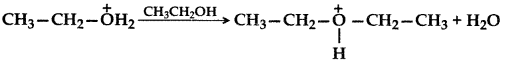1. Out of AICI3 and NaCl, which is more effective in causing coagulation of a negative sol and why?
Answer: AlCL3, because Al3+ has higher charge than Na+ ion. Higher the charge, more effective it will be for coagulation.
2. What is the type of charge on Agl colloidal sol formed when AgNO3 solution is added to KI solution?
Answer: Negative charge because negatively charged I– ions will get adsorbed on Agl.
3. What is the effect of temperature on adsorption?
Answer: Adsorption decreases with increase in temperature because adsorption is exothermic process.
4. Write the dispersed phase and dispersion medium of paints.
Answer: Solid is dispersed phase and liquid is dispersion medium.
5. Write the dispersed phase and dispersion medium of butter.
Answer: Liquid is dispersed in solid in solidified butter. So, liquid is dispersion medium and solid is dispersed phase.
6. Out of BaCl2 and KC1, which one is more effective in causing coagulation of a negatively charged colloidal Sol? Give reason.
Answer: BaCl2 will be more effective because Ba2+has higher charge than K+.
7. A delta is formed at the meeting point of sea water and river water. Why?
Answer: Muddy river water is a colloidal solution, which gets coagulated by electrolytes present in sea water.
8. Physisorption is reversible while chemisorption is irreversible. Why?
Answer: In physisorption, forces of attraction are weak and no new substance is formed, therefore, it is reversible, whereas in chemisorption, new substances are formed, therefore, it is irreversible.
Short Answer Type Questions [II] [3 Marks]
9. Define adsorption with an example. Why is adsorption exothermic in nature?
Write the types of adsorption based on the nature of forces between adsorbate and adsorbent.
Answer: The accumulation of molecular species on the surface rather than bulk of solid or liquid, e.g. 02, H2, CO, NH3 , Cl2, SO3, taking in a close vessel on charcoal. Adsorption is exothermic because of force of attraction between adsorbate and adsorbent.
Types of adsorption:
(i) Physical adsorption (Weak van der Waals’ forces)
(ii) Chemical adsorption (Due to formation of covalent bonds)
10. Differentiate between the following:
(i) Solution and colloid
(ii) Homogeneous catalysis and Heterogeneous catalysis
(iii) O/W emulsion and W/O emulsion.
Answer: (i) Solution is homogeneous, whereas colloid is heterogeneous.
(ii) In homogeneous catalysis, catalyst and reactants are in the same physical states, whereas in heterogeneous catalysis, reactants and catalyst are in different physical states.
(iii) Oil is dispersed phase and water is dispersion medium in O/W emulsion, whereas water is dispersed phase and oil is dispersion medium in W/O emulsion.
11. Give reasons of the following observations:
(i) Physisorption decreases with increase in temperature.
(ii) Addition of alum purifies the water.
(iii) Brownian movement provides stability to the colloidal solution.
Answer: (i) It is because of weak van der Waals’ forces of attraction which decrease with increase in temperature.
(ii) Alum coagulates mud particles and purify water.
(iii) Brownian movement does not allow colloidal solution to settle down due to constant zig-zag motion.
12.Give reasons for the following observations:
(i) Leather gets hardened after tanning.
(ii) Lyophilic sol is more stable than lyophobic sol.
(iii) It is necessary to remove CO when ammonia is prepared by Haber’s process.
Answer: (i) It is due to coagulation. When animals hide, which is positively charged is soaked in tannin, which contains negatively charged particles, mutual coagulation takes place.
(ii) It is due to more force of attraction between dispersed phase and dispersion medium in Lyophilic sol than Lyophobic sols.
(Hi) CO will react with Fe to form Fe(CO)5 and acts as catalytic poison (inhibitor), therefore, it must be removed.
13. Write any three differences between Physisorption and Chemisorption.
Answer:
14. Give reasons for the following observations:
(i) A delta is formed at the meeting point of sea water and river water.
(ii) NH3 gas adsorbs more readily than N2 gas on the surface of charcoal.
(iii) Powdered substances are more effective adsorbents.
Answer: (i) Muddy river water is a colloidal solution, gets coagulated by electrolytes present in sea water and delta is formed.
(ii) NH3 has more van der Waals’ forces of attraction than N2,, as NH3 is polar and can form H-bonds. Therefore, it can get adsorbed to more extent than N2.
(iii) Powdered substance has more surface area, therefore, it gets adsorbed to more extent. Greater the surface area, more will be the extent of adsorption.
2014
Very Short Answer Type Questions [1 Mark]
15. Give one example each of ‘oil in water’ and ‘water in oil’ emulsion.
Answer: Milk is an example of ‘oil in water’, whereas butter (liquid) is an example of ‘water in oil’ emulsion.
16. Give one example each of ‘sol’ and ‘gel’.
Answer: starch in water is an example of ‘sol’, whereas cheese is an example of ‘gel’.
17. Give one example each of lyophobic sol and lyophilic sol.
Answer: Starch in water is lyophilic sol, whereas As2S3 in water is lyophobic sol.
18.Why is adsorption always exothermic?
Answer: Δ G = Δ H – TΔ S
Δ S = -ve only if Δ H is -ve,
i.e. adsorption is always exothermic process.
19. What are the dispersed phase and dispersion medium in milk?
Answer: Liquid (protein, lactose, fat) is dispersed phase and liquid (water) is dispersion medium.
20. What is the effect of temperature on chemisorption?
Answer: Chemisorption first increases and then decreases with increase in temperature.
21.What type of forces are responsible for the occurrence of physisorption?
Answer: Weak van der Waals’ forces of attraction are responsible for the occurrence of physisorption.
22. Name the temperature above which the formation of micelles takes place.
Answer: Kraft temperature.
23. Based on the type of dispersed phase, what type of colloid is micelles?
Answer: Associated colloids.
24. Define the term ‘Tyndall effect’.
Answer: When light passes through a colloidal solution, its path becomes clearly visible due to scattering of light by colloidal particles. This is called ‘Tyndall effect’.
25. What is meant by ‘shape-selective catalysis’ of reactions?
Answer: Shape-selective catalysis: The catalysis which depends upon the pore structure of the catalyst and molecular size of reactant and product molecules is called shape-selective catalysis, e.g. Zeolites are shape-selective catalysts due to their honeycomb structure. ZSM-5 (Zeolite Sieve of Molecular porosity 5) is used to convert methanol into gasoline (petrol).
26. What is meant by dialysis.
Answer: It is a process of removing dissolved substance from a colloidal solution by means of diffusion through suitable membranes.
Short Answer Type Question [II] [3 Marks]
27. What are emulsions? What are their different types? Give one example of each type.
Answer: Liquid in liquid colloidal dispersions are called emulsions.
Types of emulsions: (i) oil in water emulsions (it) water in oil emulsions.
Milk is an example of oil in water, whereas liquefied butter is an example of water in oil emulsion.
28. (a) In reference to Freundlich adsorption isotherm write the expression for adsorption of gases on solids in the form of an equation.
(b) Write an important characteristic of lyophilic sols.
(c) Based on type of particles of dispersed phase, give one example each of associated colloid and multimolecular colloid.
Answer: (a) log x/m = log k + 1/n log P
where x = mass of the gas adsorbed, m = mass of adsorbent at equilibrium pressure P, k and n are constants.
(b) Lyophilic sols are stable and reversible.
(c) Soaps and detergents form associated colloids, whereas As2S3 and gold sol form multimolecular colloids.
29. (a) Write the expression for the Freundlich adsorption isotherm for the adsorption of gases on solids, in the form of an equation.
(b) What are the dispersed phase and dispersion medium of butter?
(c) A delta is formed at the meeting place of sea and river water. Why?
Answer: (a) Refer Ans. to Q.28 (a).
(b) Refer Ans. to Q.5.
(c) Refer Ans. to Q.14 (i).
30. Define the following terms:
(i) Adsorption (ii) Peptization (iii) Sol.
Answer: (i) Adsorption: When concentration of solute is different at surface than bulk, it is called adsorption.
(ii) Peptization: It is a process of converting freshly prepared precipitate into colloidal solution in the presence of stabilizing agent.
(iii) Sol: When a solid is dispersed in liquid, it is called sol.
31. Giving appropriate examples, explain how the two types of processes of adsorption (physisorption and chemisorption) are influenced by the prevailing temperature, the surface area of the adsorbent and the activation energy of the process?
Answer: Effect of temperature: Physisorption decreases with increase in temperature, where chemisorption first increases, then decreases with increase in temperature. Surface area: Greater the surface area, greater is the physisorption and chemisorption.
Activation energy: In physisorption, no appreciable activation energy is needed, whereas in chemisorption, sometimes high activation energy is needed.
2013
Very Short Answer Type Questions [1 Marks]
32. Of physisorption or chemisorption, which has a higher enthalpy of adsorption?
Answer: Chemisorption because it has high enthalpy of adsorption.
33. Which aerosol depletes ozone layer?
Answer: Chlorofluorcarbons (CFC)
34. What is especially observed when a beam of light is passed through a colloidal solution?
Answer: Its path becomes clearly visible due to scattering of light. It is called ‘Tyndall effect’.
Short Answer Type Questions [I] [2 Marks]
35. Write the dispersed phase and dispersion medium of the following colloidal system:
(i) Smoke (ii) Milk
Answer:
36. What are lyophilic and lyophobic colloids? Which of these sols can be easily coagulated on the addition of small amounts of electrolytes?
Answer: Difference between lyophilic sols and lyophobic sols:
Lyophobic sols can be easily coagulated on addition of small amount of electrolytes.
37. What is the difference between oil/water (O/W) type and water/oil (W/O) type emulsions? Give an example of each type.
Answer: When oil is dispersed in water, it is called oil in water emulsion, e.g. milk. When water is dispersed in oil, it is called water in oil emulsion, e.g. butter.
38. What is the difference between multimolecular and macromolecular colloids? Give one example of each.
Answer: Differences between Multimolecular and Macromolecular colloids.

39.(a) What happens when a freshly precipitated Fe(OH)3 is shaken with water containing a small quantity of FeCl3?
(b) Why is a finely divided substance more effective as an adsorbent?
Answer: (a) Colloidal solution of Fe(OH)3 is formed. FeCl3acts as peptizing agent.
(b) It is due to greater surface area and it has more active sites. Therefore, extent of adsorption is more.
40. Write two differences between lyophobic and lyophilic sols. Give one example of each type of sol.
Answer: Refer Ans. to Q.36.
41. What is special about the following terms:
(i) Kraft Temperature (ii) Sorption
Answer: (i) Kraft Temperature (Tk): Micelles form ionic surfactants (surface active agents like soaps and detergents) only above a certain temperature called Kraft temperature.
(ii) Sorption: When adsorption and absorption take place simultaneously, it is called sorption, e.g. dyeing of cotton fabrics. The dye is adsorbed on the surface of cotton fibre but after it is dyed, the fibre has dye uniformly throughout, i.e. absorbed.
42. Write the dispersed phase and dispersion medium of the following colloids:
(i) Cheese (ii) Fog
Answer: (i) Dispersed phase is liquid (water), dispersion medium is solid (cheese).
(ii) Dispersed phase is liquid (water vapours), dispersion medium is gas(air).
43. Write the differences between physisorption and chemisorption with respect to the following:
(i) Specificity (ii) Temperature dependence
(iii) Reversibility and (iv) Enthalpy change
Answer: Differences between Physical and Chemical adsorptions.
44. Describe a chemical method each for the preparation of the sols of sulphur and platinum in water.
Answer:Sulphur sol can be prepared by oxidation of H2S with the help of cone. HNOg.![]()
Bredig’s Arc Method:
Platinum (Pt) sol can be prepared by Bredig’s arc method. Two Pt metal electrodes are takeri through which high voltage is passed so that metal gets vaporised. When vapours are condensed, colloidal solution of Pt in water is obtained.
Short Answer Type Question [II] [3 Marks]
45. What are the characteristics of the following colloids? Give one example of each.
(i) Multimolecular colloids
(ii) Lyophobic sols
(iii) Emulsions
Answer: (i) Multimolecular colloids: Colloidal particles consist of aggregates of small atoms or molecules. For example, As2S3 sol.
(ii) Lyophobic sols: There is no affinity between dispersed phase and dispersion medium. They are prepared by indirect methods. For example, Fe(OH)3sol and gold sol.
(iii) Emulsions: When liquid is dispersed in liquid, it is called emulsion, e.g. milk and butter (liquid).
46. Define the following terms giving an example of each:
(i) Associated colloids (ii) Lyophilic sol (iii) Adsorption
Answer: (i) Associated colloids: These colloids behave like electrolytes in low
concentration but at high concentrations, due to aggregated particles, form colloidal solution.
They form ions in low’ concentration but ions get aggregated in high concentration, e.g. soaps and detergents (surface active agents).
(ii) Lyophilic sol: There is a force of attraction between dispersed phase and dispersion medium. They are easily prepared by shaking dispersed phase with dispersion medium. For example, gum and starch in water form lyophilic sols.
(iii) Adsorption: The existence of a substance at a surface in different concentration than in the adjoining bulk is called adsorption, e.g. 02, H2, CO, Cl2, NH3 or S02 get adsorbed on the surface of charcoal.
47. Define the following terms writh an example in each case:
(i) Macromolecular sol (ii) Peptization (iii) Emulsion
Answer: (i) Macromolecular sol: Dispersed particles are large molecules (usually polymers). They have force of attraction between dispersion medium and dispersed phase, e.g. albumin in water, proteins and stajrch in water, cellulose nitrate in alcohol (collodion).
(ii) Peptization: The conversion of precipitate into colloidal solution in presence of peptizing agent is called peptization. Peptizing agent is generally an electrolyte. In this process, dispersion medium (liquid) breaks up the precipitate into colloidal state.
(iii) Emulsion: Emulsion is liquid dispersed in liquid, e.g. milk.
48. (a) Which will adsorb more gas, a lump of charcoal or its powder and why?
(b) Describe the preparation of the following colloidal solutions. Name the method used in each case:
(i) Silver sol (ii) Sulphur sol
Answer: (a) Charcoal powder will adsorb more gas because it has more surface area.
(b) (i) Bredig’s Arc method: Take silver electrodes in the form of electric arc in water and pass high voltage through it. A lot of heat will be generated which will vaporise silver metal into vapours which on condensing form silver sol.
(ii) Oxidation: Pass H2S gas through aqueous solution of nitric acid. Colloidal solution of sulphur will be obtained.![]()
49. (a) What are the two types of emulsions and how do they differ from one another? Give one example of each.
(b) Which one of the following electrolytes is most effective for the coagulation of Fe(OH)3 sol and why?
NaCl, Na2SO4, Na3P04
Answer: (a) (i) Oil in water: When oil is dispersed in water, it is called oil in water emulsion, e.g. milk.
(ii) Water in oil: When water is dispersed in oil, it is called water in oil emulsion, e.g. butter in liquid state.
(b) Fe(OH)3 is positively charged colloid, therefore, Na3P04 will be most effective for coagulation. It is because P04 ions are negatively charged and have highest charge among these electrolytes.
2012
Very Short Answer Type Questions [1 Mark]
50. Define peptization?
Answer: Refer Ans. to Q.47 (ii).
51. What is meant by ‘shape-selective catalysis’?
Answer: Refer Ans. to Q.25.
52. How is sol different from an emulsion?
Answer: Sol: When solid is dispersed in liquid, it is called sol.
Emulsion: When liquid is dispersed in liquid, it is called emulsion.
53. Why is adsorption phenomenon always exothermic?
Answer: Refer Ans. to Q.18.
Short Answer Type Questions [i] [2 Marks]
54. Explain the following terms giving one example for each:
(i) Micelles (ii) Aerosol
Answer: (i) Refer Ans. to Q.58 (iii).
(ii) Refer Ans. to Q.58 (i).
55. Name the two groups into which phenomenon of catalysis can be divided. Give an example of each group with the chemical equation involved.
Answer: (i) Homogeneous catalysis
(ii) Heterogeneous catalysis
(i) Homogeneous catalysis:![]()
(ii) Heterogeneous catalysis:![]()
56. What is meant by coagulation of a colloidal solution? Describe any three methods by which coagulation of lyophobic sols can be carried out.
Answer: Coagulation: The process of converting a colloidal solution into precipitate is called coagulation.
Coagulation of lyophobic sols can be carried out by
(i) Electrophoresis: The colloidal particles get attracted towards oppositely charged electrodes, get discharged and coagulated.
(ii) Mutual coagulation: When negatively charged colloid is mixed with positively charged colloid, mutual coagulation takes place.
(iii) Boiling: The charge on colloidal particles is reduced due to collision of colloidal particles with particles of dispersion medium which leads to coagulation.
Short Answer Type Question [II] [3 Marks]
57. Explain what is observed when
(i) an electric current is passed through a sol.
(ii) a beam of light is passed through a sol.
(iii) an electrolyte (say NaCl) is added to ferric hydroxide sol.
Answer: (i) The colloidal particles get attracted towards one of the electrodes depending upon charge on colloidal particles.
(ii) The path of light becomes clearly visible due to scattering of light by colloidal particles.
(iii) The sol gets coagulated.
58. Explain the following terms giving a suitable example for each:
(i) Aerosol (ii) Emulsion
(iii) Micelles
Answer:
(ii) Refer Ans. to Q.47 (iii).
(iii) Micelles: Soaps and detergents consist of lyophilic and lyophobic parts which associate together to form micelles.
59. Differentiate among a homogeneous solution, suspension and a colloidal
solution, giving a suitable example of each.
Answer: Homogeneous solution: The particle size is less than 1 nm. Particles cannot be seen even with powerful microscope. It is transparent, e.g. salt solution. Suspension: It is heterogeneous, particle size is greater than 1000 nm. Particles can be seen with naked eye. It is opaque, e.g. chalk powder in water. Colloidal solution: It appears to homogeneous but actually heterogenous. Particles can be seen with powerful microscope. It is translucent, e.g. milk.
60. Present a classification of colloids where dispersion medium is water. State the characteristics and one example of each of these classes.
Answer: (i) Sol: When solid is dispersed in water, it is called sol, e.g. starch sol and gold sol.
(ii) Emulsion: When liquid is dispersed in water, it is called emulsion, e.g. milk.
(iii) Foam: When gas is dispersed in water, it is called foam or froth, e.g. lemonade froth.
61. Define each of the following terms:
(i) Micelles (ii) Peptization
(iii) Electrophoresis
Answer: (i) Refer Ans. to Q.58 (iii).
(ii) Refer Ans. to Q.47 (ii).
(iii) Electrophoresis: The movement of colloidal particles towards positive or negative electrode in an electric field is called electrophoresis. It occurs due to the presence of positive or negative charge on colloids.
62. Write three distinct features of chemisorptions which are not found in
physisorptions.
Answer: Refer Ans. to Q.13.
63. How are the two types of emulsions different from one another? Give suitable examples to justify the difference. State two applications of emulsions.
Answer: When oil is dispersed in water, it is oil in water emulsion, e.g. milk. It w-e add water, it mixes up showing that it is oil in water emulsion. When water is dispersed in oil, it is called water in oil emulsion, e.g. in butter, if we add oil, it will get mixed up”, so, it is water in oil emulsion.
Applications:
(i) Emulsifying properties of soaps and detergents are used in washing clothes, crockery, etc.
(ii) Digestion of fats in the intestine is facilitated by emulsification.
2011
Very Short Answer Type Questions [1 Mark]
64. What is meant by ‘shape-selective catalysis’?
Answer: Refer Ans. to Q.25.
65. Define electrophoresis.
Answer: Refer Ans. to Q.61 (iii).
66. What are lyophobic colloids? Give one example for them.
Answer: The colloids in which there is least force of attraction between dispersed phase and dispersion medium e.g. As2S3 sol.
67. Why is a finely divided substance more effective as an adsorbent?
Answer: Refer Ans. to Q.39 (b).
68. What is meant by chemisorption?
Answer: Chemisorption: If the forces holding the adsorbate are as strong as in chemical bonds, the adsorption process is known as chemical adsorption or chemisorption.
69. What is the basic difference between adsorption and absorption?
Answer: Adsorption is a selective absorption when cone, of solute is more at the surface and less in the bulk. Absorption is a process in which cone, of solute is uniform throughout.
Short Answer Type Questions [I] [2 Marks]
70. Define the following terms giving an example of each:
(i) Emulsion
(if) Hydrosol
Answer: (i) Refer Ans. to Q.45 (iii).
(ii) When solid is dispersed in water, it is called hydrosol, e.g. starch dispersed in water.
71. Write four distinguishing features of operative between chemisorption and physisorption.
Answer: Refer Ans. to Q.43.
72. Define the following terms:
(i) Aerosol (ii) Coagulation of colloids
Answer: (i) Refer Ans. to Q.58 (i).
(ii) The process of converting colloidal solution into precipitate is called coagulation of colloids.
Short Answer Type Questions [II] [3 Marks]
73. Define each of the following terms:
(i) Micelles (ii) Peptization
(iii) Electrophoresis
Answer: Refer Ans. to Q.61.
74. Explain how the phenomenon of adsorption finds application in each of the following processes:
(i) Production of vacuum (ii) Heterogeneous catalysis (iii) Froth Floatation process
Answer: (i) Production of vacuum: The high vacuum can be created by adsorbing remaining gases by charcoal after removing air by vacuum pump.
(ii) Heterogeneous catalysis: When the catalyst forms a separate phase (usually a solid phase) from reactants, it is said to be heterogeneous and the catalysis is heterogeneous catalysis. The example of heterogeneous catalysis is manufacture of H2S04 in contact process using V2O2 as catalyst. Usually, in a heterogeneous catalysis, the reactants are gases and reaction starts from the surface of the solid catalyst. This is the reason why heterogeneous catalysis is also called ‘surface catalysis’.
(iii) Froth Floatation Process: The principle of froth floatation process is that sulphide ores are preferentially wetted by (adsorbed on) the pine oil and frothing agent, whereas the gangue particles are wetted by the water.
75. Classify colloids where the dispersion medium is water. State their characteristics and. write an example of each of these classes.
Answer: Refer Ans. to Q.60.
76. Explain the following terms:
(i) Electrophoresis
(ii) Dialysis
(iii) Tyndall effect
Answer: (i) Electrophoresis: The movement of colloidal particles towards positive or negative electrode in an electric field is called electrophoresis. It occurs due to the presence of positive or negative charge on colloids.
(ii) Dialysis: It is a process of purification of sols containing electrolyte by keeping the sol in a bag made up of parchment or cellophane and suspending the bag in pure water. Small molecules or ions can pass through the membrane, but the sol is retained.
(iii) Refer Ans. to Q.24.
77. Explain what is observed when
(i) an electric current is passed through a sol.
(ii) a beam of light is passed through a sol.
(iii) an electrolyte (say NaCl) is added to ferric hydroxide sol.
Answer: (i) The colloidal particles get attracted towards one of the electrodes depending upon the charge on colloidal particles.
(ii) The path of light becomes clearly visible due to scattering of light by colloidal particles.
(iii) The sol gets coagulated.
2010
Very Short Answer Type Questions [1 Mark]
78. Name the two types of adsorption phenomenon.
Answer: (i) Physical adsorption (physisorption)
(ii) Chemical adsorption (Chemisorption)
79. What is the basic difference between adsorption and absorption?
Answer: Refer Ans. to Q.69.
80. What is meant by Reversible sols.
Answer: Reversible sols: Those sols in which dispersion medium can be separated from the dispersed phase (e.g. by evaporation), the sol can be made again by remixing with dispersion medium, e.g. lyophilic sols.
81. What is an emulsion?
Answer: Refer Ans. to Q.45 (iii).
Short Answer Type Questions [I] [2 Marks]
82. Describe the following:
(i) Tyndall effect (ii) Shape-selective catalysis.
Answer: (i) Refer Ans. to Q.24.
(ii) Refer Ans. to Q.25.
83. What is meant by coagulation of a colloidal solution? Name any method by which coagulation of lyophobic sols can be carried out.
Answer: Refer Ans. to Q.56.
84. What are emulsions? State one application of emulsification.
Answer: Liquids dispersed in liquids are called emulsions. Cleansing action of soaps and detergents is based on emulsification.
85. What is the difference between a colloidal solution and emulsion? What is the role of emulsifier in forming emulsion?
Answer: Colloidal solution is a solution in which either solid is dispersed in solid, liquid or gases or liquid is dispersed in solid, liquid or gases or gases are dispersed in solids or liquids.
Emulsions are colloidal solution in which liquid is dispersed in liquid. Emulsifier stabilizes the emulsion.
86. Define the following:
(i) Peptization (ii) Reversible sols.
Answer: (i) Refer Ans. to Q.30 (ii).
(ii) Refer Ans. to Q.80.
Short Answer Type Questions [II] [3 Marks]
87. State what is observed when
(i) an electrolyte, NaCl, is added to hydrated ferric oxide sol.
(ii) an electric current is passed through a colloidal solution.
(iii) a beam of light is passed through a colloidal solution
Answer: (i) The sol gets coagulated.
(ii) The colloidal particles get attracted towards one of the electrodes depending upon charge on colloidal particles.
(iii) The path of light becomes clearly visible due to scattering of light by colloidal particles.
88. What is the difference between multimolecular and macromolecular colloids? Give one example of each type. How are associated colloids different from these two types of colloids?
Answer: (i) Multimolecular colloids: Those colloids in which small atoms or molecules combine together to form the particles of colloidal size are called multimolecular colloids, e.g. gold sol, sulphur sol, As2S3 sol, etc.
(ii) Macromolecular colloids: Those colloids in which dispersed particles are macromolecules (bigger molecules), i.e. polymers are called macromolecular colloids, e.g. starch in water, gum in water.
(iii) Associated colloids: Those colloids which behave as strong electrolyte at low concentrations, but show colloidal properties at higher concentration due to the formation of aggregated particles of colloidal dimensions. Such substances are called associated colloids, e.g. soaps and synthetic detergents belong to this class. Micelles are the aggregated particles or associated colloids in solution.
89. Discuss the effect of pressure and temperature on the adsorption of gases on solids. Describe the application of adsorption in controlling humidity.
Answer: Adsorption is a process in which the concentration of solute is more at the surface and less in the bulk. It is called positive adsorption.
Physical adsorption of a gas on solid decreases with increase in temperature and increases with increase in pressure.
Chemical adsorption first increases and then decreases with increase in temperature.
Chemical adsorption first increases and then becomes independent of pressure with increase in pressure. x/m represents the extent of adsorption where x is the mass of adsorbate and m is mass of adsorbent.
90. How are the following colloids different from each other in respect of their dispersion medium and dispersed phase? Give one example of each.
(i) Aerosol
(ii) Emulsion
(iii) Hydrosol
Answer:
91. Write three distinct features of chemisorptions which are not found in physisorptions.
Answer: Refer Ans. to Q. 13.
92. Write four features of chemisorptions which are not found in physisorptions. Illustrate your answer with suitable examples.
Answer: Refer Ans. to Q.43.
2009
Very Short Answer Type Questions [1 Mark]
93. What is an emulsion?
Answer: Refer Ans. to Q.45 {iii).
94. What is the ‘coagulation’ process?
Answer: The conversion of a colloidal solution into precipitate is called coagulation. In other words, the process of settling of colloidal particles is called coagulation.
95. Define the term Tyndall effect.
Answer: Refer Ans. to Q.24.
Short Answer Type Questions [II] [3 Marks]
96. State what is observed when
(i) an electrolyte, NaCl, is added to hydrated ferric oxide sol.
(ii) an electric current is passed through a colloidal solution.
{iii) a beam of light is passed through a colloidal solution
Answer: Refer Ans. to Q.87.
97. What is the difference between multimolecular and macromolecular colloids? Give one example of each type. How are associated colloids different from these two types of colloids?
Answer: Refer Ans. to Q.88.
98. How are the following colloids different from each other in respect of their dispersion medium and dispersed phase? Give one example of each.
(i) Aerosol {ii) Emulsion (iii) Hydrosol
Answer: Refer Ans. to Q.90.
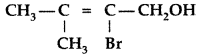
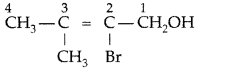
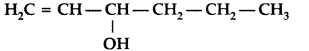
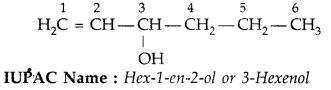
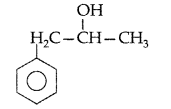
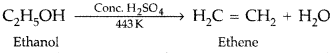
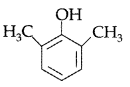
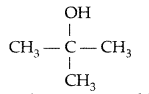
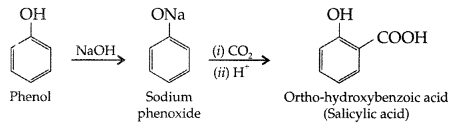
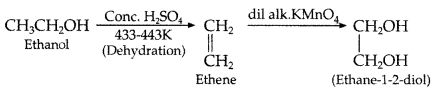
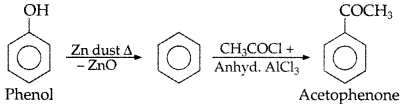
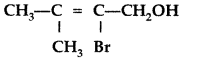
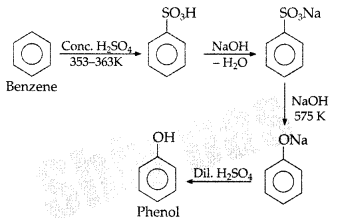
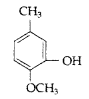
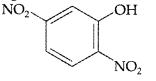
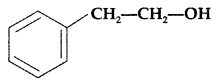
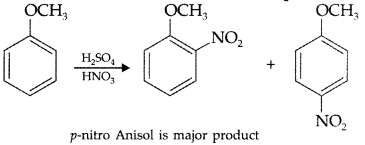
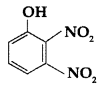
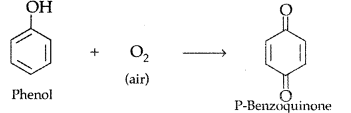
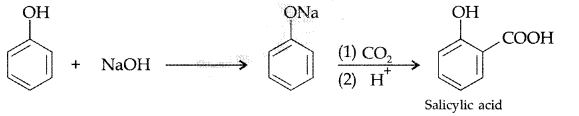
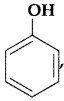 , which one is more acidic? (Comptt. All India 2016)
, which one is more acidic? (Comptt. All India 2016)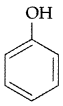 is more acidic, as Phenoxide formed is more stabilized by Resonance.
is more acidic, as Phenoxide formed is more stabilized by Resonance.